The millions of Americans who suffer from moderate to severe sleep apnea now have an alternative to CPAP (continuous positive airway pressure) treatment with the Inspire Procedure.

Upper airway stimulation, also known as hypoglossal nerve stimulation, is an entirely different way to treat sleep apnea. It has a high success rate and minimal complications, making it an excellent choice for many patients.
Until recently, moderate to severe sleep apnea sufferers have had few treatment options, and they each came with significant challenges. Treatment with a CPAP machine has been the gold standard for treating sleep apnea, but it is not practical for many patients. Traditional surgical options that target the soft-palate tissue have only moderate success rates and often cause a great deal of pain and discomfort.
With a surgically implanted device that painlessly stimulates the upper airway, sleep apnea patients can maintain normal breathing patterns and finally be free from their CPAP machine.
What is Obstructive Sleep Apnea (OSA)?
Obstructive sleep apnea is a common sleep disorder that disrupts normal breathing. With OSA, the air passages narrow as throat muscles relax during sleep, which causes a constant stopping and starting of breathing. One of the most common signs of sleep apnea is snoring.
When breathing is disrupted during sleep, your body is deprived of oxygen which has negative health effects on many different parts of the body including the heart and brain. In addition to snoring, some OSA patients experience other symptoms like awakening during the night while gasping or choking, daytime sleepiness, high blood pressure, headaches, weight gain, depression or anxiety. Sleep apnea can also have a negative impact on a bed partner’s sleep quality. If you are experiencing any combination of the symptoms above, it is helpful to talk to your Primary Care Provider. You can find PCPs in your area here.
Causes of Obstructive Sleep Apnea?
These conditions may put you at a higher risk of developing sleep apnea:
- High blood pressure
- Diabetes
- Smoking
- Obesity
- Narrowed airways or chronic nasal congestion
- Male gender
How the Inspire Device Works?
Upper airway stimulation involves having a medical device surgically implanted under the skin, in the chest area. The device remains in that location, indefinitely, and is controlled by a remote.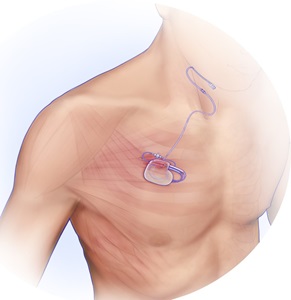
The Inspire device monitors breathing and with each breath, sends a signal to the hypoglossal nerve that causes the throat muscles to open instead of collapse, thus preventing the tongue from blockng the airway. The stimulation is mild and patients remain asleep while the device is working.
This innovative sleep apnea surgery has a significant impact on snoring and other symptoms, with 80% to 90% of patients achieving normal sleep patterns.
Who is a candidate?
Successful candidates for hypoglossal nerve stimulation meet these requirements:
- Moderate to severe sleep apnea
- Age 22+
- BMI less than 35
- Unable to tolerate CPAP treatment
- Successful DISE test result
DISE (drug-induced sleep endoscopy) test
Before having surgery to implant the Inspire device, patients must undergo a DISE test. This brief sleep test is conducted in a surgical setting to observe how the throat moves while asleep. Most OSA patients experience soft-palate movement in an anterior to posterior direction while sleeping, and these patients usually have success with the upper airway device. A small portion of patients (approximately 20%) experience lateral soft-palate movement, and are not candidates for the hypoglossal nerve stimulator.
What to Expect During the Procedure
This is an outpatient surgical procedure. You will receive general anesthesia so that you do not feel pain or remember the surgery.
Two small incisions are made:
- One incision in the chest, near the collarbone, to place the battery device
- One incision under the chin to place a wire that connects with the hypoglossal nerve
Once the device is implanted and connected to the stimulation wire, the incisions are closed. Patients are able to go home the same day.
After the Procedure
The first step is to allow the surgical incisions to heal completely, ensuring the device will remain in the correct location. Healing usually takes about a month, and the device remains inactive during this time.
inactive during this time.
One month after surgery, patients are seen by a sleep-medicine provider to activate the device. After this appointment, you will be able to control the device with a remote. The device is turned on at bedtime and turned off in the morning. While asleep, the device monitors sleep patterns and stimulates the hypoglossal nerve to ensure normal breathing.
The device may require periodic adjustments to achieve and maintain the proper level of nerve stimulation.
Frequently Asked Questions (FAQs)
Most patients experience only minor pain or discomfort following the procedure.
Patients should limit heavy activity that involves the upper body and neck for about 4 weeks. Patients are advised to not participate in sports or exercise activity during that time.
The incisions heal for about 4 weeks after surgery, and then the device is activated.
The battery lasts for about 11 years. When it needs to be replaced, you will need a minor surgical procedure to change the battery.
Most normal activities, including swimming and other water activities, are not affected by the device. Limitations are similar to those for patients with a pacemaker device. You can go through metal detectors at the airport but should alert TSA agents about your device.
While the device is implanted, you will not be able to have a magnetic resonance imaging (MRI) exam of the chest or abdomen. You are able to have an MRI exam of other areas of the body, including arms, legs, neck and brain. If you experience a condition where a physician needs to view areas of your body that should not be accessed by MRI technology, there are other options including CT scan.
Nonsurgical treatment options include weight loss or dental devices. These may or may not have an impact and are often only somewhat effective for improving symptoms of mild sleep apnea.
Traditional surgery for sleep apnea involves removing tissue of the soft palate or moving the jaw forward. These procedures have only moderate success rates and some are very painful. The upper airway stimulation procedure is more successful and involves little, if any, pain.
Any surgical procedure has a risk of bleeding or infection. There is a slight risk of damage to the nerve that controls the tongue, but this is not common.
Contact Us for a Physician Referral
Fill out the form below to be connected with a Memorial Hermann-affiliated surgeon. Patient criteria for the Inspire Procedure are as follows:
- Diagnosis of Obstructive Sleep Apnea
- Struggle with CPAP
- Have had a Sleep Study within the last 2 years (preferred). If not, your physician will need to order one to have an accurate understanding of your sleep apnea.